(WKBN)- The U.S. Food & Drug Administration announced on Tuesday in a press release that some Continuous Positive Airway Pressure (CPAP) machines are subject to an emerging safety issue.
According to the FDA, the Philips Respironics’ DreamStation 2 CPAP machine was having thermal issues that caused patient injuries. The machine is used for the treatment of obstructive sleep apnea.
The FDA recently received medical device reports (MDRs) associated with issues, such as fire, smoke, burns and other signs of overheating while using the machine. The FDA is in discussions with the company about mitigation strategies for this safety issue and will update the public accordingly.
The full article is available at ozarksfirst.com.
(Story by Michael Reiner, WKBN, found at ozarksfirst.com)






 Man Charged with Murder at Apartment Complex Near Branson
Man Charged with Murder at Apartment Complex Near Branson
 Elevate Branson to put First Tiny Homes on Foundations
Elevate Branson to put First Tiny Homes on Foundations
 Building of Branson Business Recently Closed Burns
Building of Branson Business Recently Closed Burns
 Front Sweeps Through Bringing Variety of Weather, Elevated Fire Risk
Front Sweeps Through Bringing Variety of Weather, Elevated Fire Risk
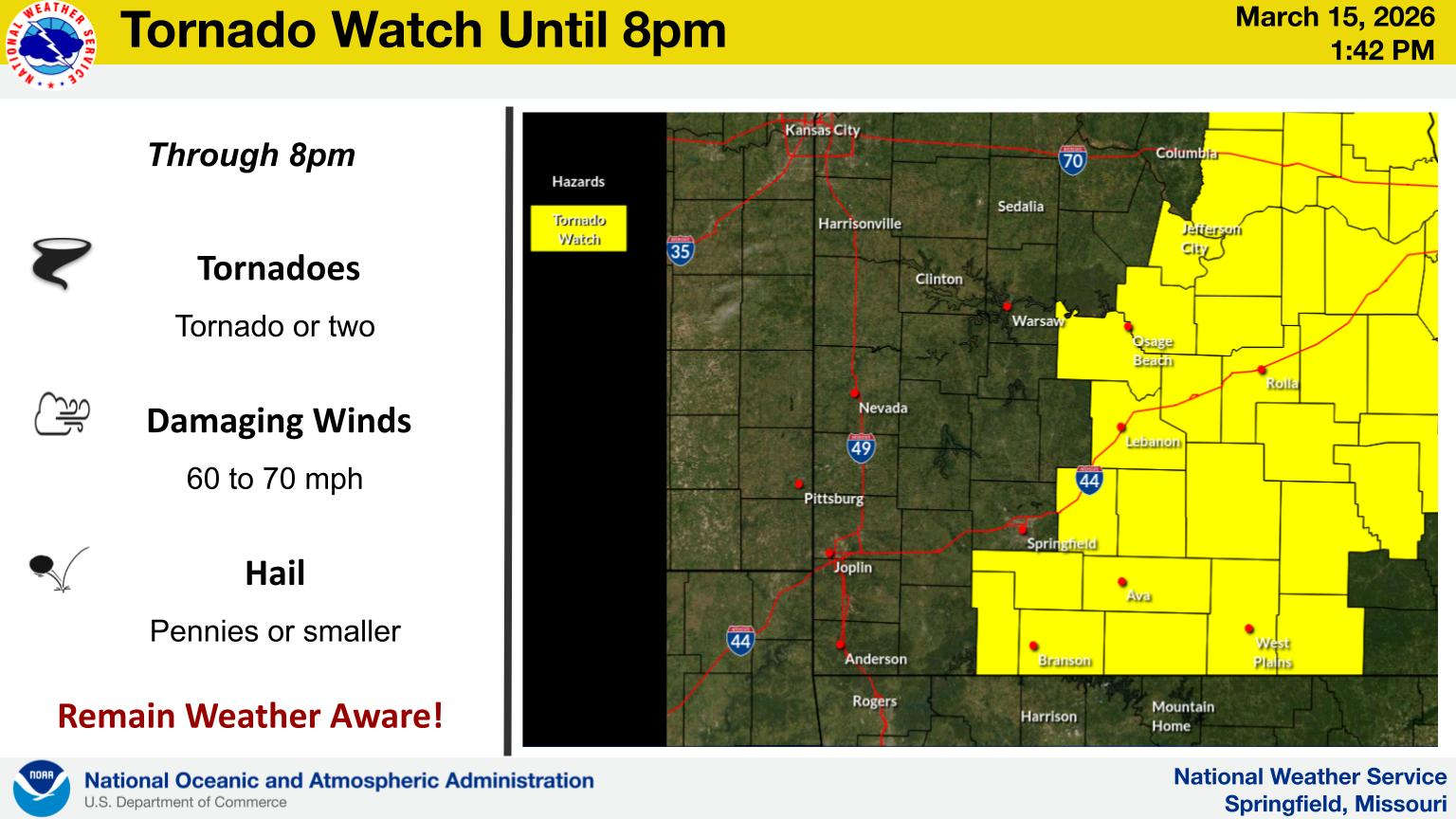 Major Weather Change Today, Precipitation Chances Drop, Strong Winds Expected UPDATE 2
Major Weather Change Today, Precipitation Chances Drop, Strong Winds Expected UPDATE 2